The mole
|
The mole is defined as the amount of substance which contains the same number of particles (atoms, ions or molecules) as there are atoms in exactly 12g of carbon.
1 mole is equal to 6.02 × 10^23 particles - This is known as Avogadro’s number. Just like we say 12 eggs is 1 dozen, 6.02 × 10^23 particles is 1 mole. It is simply a term for a number. 1 dozen = 12 items 1 mole = 6.02 × 10^23 particles |
|
|
Introduction to Moles summary:
A mole is a term for a specific number of things
|
|
|
This clip explains how you can use moles to count atoms using mass. An analogy used is if someone orders 10, 000 jellybeans at a candy store. Instead of having to count them ALL out, you could just figure out the mass and weigh out that amount.
The same principle can be applied to moles. To figure out how much 1 mole of an atom is in grams, look at the periodic table for the molar mass number. For example, 1 mole of sulfur weighs 32.07g, 1 mole of carbon weighs 12.01g and 1 mole of zinc weighs 65.38g. These atoms are all different in mass just like if we weighed 10,000 jellybeans, it would have a greater mass than if we weighed 10,000 M&Ms. |
If 1 mole of particles equals 6.02 x 10^23 particles
How many particles are in 0.150 mole?
6.02 x 10^23 x 0.150
= 9.03 x 10^22 particles
- 2 moles of particles = 2 x 6.02 x 10^23 particles
- 5 moles of particles = 5 x 6.02 x 10^23 particles
- 0.5 moles of particles = 0.5 x 6.02 x 10^23 particles
How many particles are in 0.150 mole?
6.02 x 10^23 x 0.150
= 9.03 x 10^22 particles
|
What amount (in moles) of each type of atom is there in:
1 mol of MgCl2 1 mol of Mg atoms and 2 mol of Cl atoms 2 mol of CaCl2 2 mol of Ca atoms and 4 mol of Cl atoms 1 mol of H2O 2 mol of H and 1 mol of O |
A sample of water contains 3.25 x 10^25 molecules. How many moles of water are there?
3.25 x 10^25 / 6.02 x 10^23 = 54.0 mol How many moles of H atoms are there? 54 x 2 = 108 moles of H atoms |
Atomic mass
The Atomic Mass gives us the average mass of one mole of atoms of that element
To find the atomic mass of an element, simply look at the periodic table. The relative atomic mass of magnesium, is 24.31. The atomic mass of carbon is 12.01. These are the masses of 1 mole of a substance.
Calculating molar masses (m)
The mass of one mole of any substance is equal to the sum of the atomic masses of its elements
|
Calculate: Molar mass (M) of H2SO4
M(H2SO4) = 2xM(H) + 1xM(S) + 4xM(O) = 2x1.01 + 1x32 + 4x16 = 98.0 g/mol |
Calculate: Molar mass (M) of CuSO4
M(CuSO4) = 1xM(Cu) + 1xM(S) + 4xM(O) = 1x63.6 + 1x32 + 4x16 = 159.6 g/mol |
Calculate: Molar mass (M) of NH4Cl
M(NH4Cl) = 1xM(N) + 4xM(H) + 1x(Cl) = 1x14 + 4x1.01 + 1x35.5 = 53.5 g/mol |
Converting mass to mole
Converting the mass of the substance (in grams) to the amount (in moles)
|
Calculate the amount (mol) in 24 grams of Carbon Dioxide (CO2)
n = m/M = 24g / 44g/mol = 0.545 mol (3sf) |
Calculate the amount (mol) in 12.2 grams of water (H2O)
n = m/M = 12.2g / 18g/mol = 0.678 mol (3sf) |
Calculate the amount (mol) in 0.160 grams of sodium hydroxide (NaOH)
n = m/M = 0.160g x 40g/mol = 4.00x10^-3 mol (3sf) |
Converting mole to mass
Converting amount (in moles) to mass (in grams)
|
Calculate the mass of 0.254 mol of SO2
First we must work out the molar mass of SO2: M(SO2) = 64.1 g/mol This means there is 64.1g in 1 mol of SO2. So how many grams are in 0.254 mol of SO2? m = n x M = 0.254 mol x 64.1 g/mol = 16.3 g (3sf) This means there are 16.3 grams of SO2 in 0.254mol |
Calculate the mass of 1.38 mol of magnesium carbonate
First we must work out the molar mass of MgCO3 M(MgCO3) = 84.3 g/mol This means there are 84.3g in 1 mol of MgCO3. So how many grams are in 1.38 mol? m = n x M = 1.38 x 84.3 g/mol = 116 g (3sf) This means there are 116 grams in 1.38 mol of MgCO3 |
mole & mass calculations for reactions
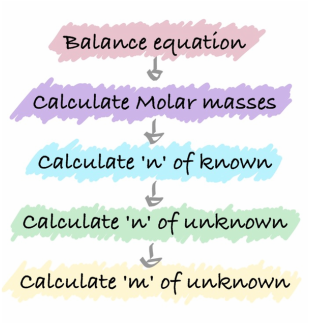
Using balanced equations to predict amount and mass of substances used
Magnesium oxide can be obtained by burning 7.20 g of magnesium ribbon in excess oxygen. Calculate the mass of oxygen used in the reaction.
|
Step 1: Write a balanced equation for the reaction
Step 2: Calculate the molar mass for the number of moles of the identified chemicals Step 3: Calculate 'n' for the chemical of known mass Step 4: Use the balanced equation to determine the mole ratio therefore the number of moles of the unknown Step 5: Using the value of n determined above from the mole ratio, use the m=n/M to determine the unknown mass |
2Mg + O2 ---> 2MgO
M(O2) = 32 g/mol M(Mg) = 24.3 g/mol n(Mg) = m/M = 7.2g / 24.3g/mol = 0.296 mol (3sf) 2Mg : 1O2 0.296 : 0.148 m(O2) = n/M = 0.148mol / 32g/mol = 4.74 g (3sf) |
Calculate the amount (in mole) of sulfuric acid required to react completely with 15.0 g of zinc
Step 1: Zn + H2SO4 ------> ZnSO4 + H2
Step 2: M(H2SO4) = 98 g/mol
M(Zn) = 65.4 g/mol
Step 3: n(Zn) = m/M
= 15.0 g / 65.4 g/mol
= 0.229 mol NB: keep this value in your calculator
Step 4: 1 Zn : 1 H2SO4
0.229 : 0.229
Step 5: m(H2SO4) = n/M
= 0.229mol / 98g/mol
= 22.5 g (3sf)
Step 1: Zn + H2SO4 ------> ZnSO4 + H2
Step 2: M(H2SO4) = 98 g/mol
M(Zn) = 65.4 g/mol
Step 3: n(Zn) = m/M
= 15.0 g / 65.4 g/mol
= 0.229 mol NB: keep this value in your calculator
Step 4: 1 Zn : 1 H2SO4
0.229 : 0.229
Step 5: m(H2SO4) = n/M
= 0.229mol / 98g/mol
= 22.5 g (3sf)
Calculate the mass of aluminium that would react completely with 17.5 g of copper(ll) chloride according to the following equation: 2Al + 3CuCl2 ---> 2AlCl3 + 3Cu
M(Al) = 27 g/mol
M(CuCl2) = 134.5 g/mol
n(CuCl2) = m/M Calculate n of the known
= 17.5g / 134.5 g/mol
= 0.130 mol In 17.5 grams of CuCl2 we have 0.130 mol
2Al : 3CuCl2 Mol ratio
0.0867 : 0.130
CuCl2 0.130 mol : 17.5 g Cross multiply: We know 3 mol produced 17.5 grams
Al 0.0867 mol : x g
0.130x = 1.52
x = 11.67
= 11.7 g (3sf)
This means that 11.7 grams of aluminium would react completely with copper(ll) chloride.
M(Al) = 27 g/mol
M(CuCl2) = 134.5 g/mol
n(CuCl2) = m/M Calculate n of the known
= 17.5g / 134.5 g/mol
= 0.130 mol In 17.5 grams of CuCl2 we have 0.130 mol
2Al : 3CuCl2 Mol ratio
0.0867 : 0.130
CuCl2 0.130 mol : 17.5 g Cross multiply: We know 3 mol produced 17.5 grams
Al 0.0867 mol : x g
0.130x = 1.52
x = 11.67
= 11.7 g (3sf)
This means that 11.7 grams of aluminium would react completely with copper(ll) chloride.
Calculate % composition of substances
To calculate the percentage of each component within a substance
Calculate the percentage composition of: MgSO4
|
First we must work out the total molar mass of the substance (remember we get individual masses from the periodic table)
M(Mg) = 24.3 g/mol M(S) = 32.1 g/mol M(O) = 16 x 4 = 64 g/mol M(MgSO4) = 24.3 + 32.1 + 64 M(MgSO4) = 120.4 g/mol |
Next, we need to divide the individual molar masses of each component (Mg, S & O) by the total molar mass of the substance (MgSO4) and multiply by 100 to convert to a percentage
% Mg = 24.3 / 120.4 x 100 = 20.2% % S = 32.1 / 120.4 x 100 = 26.7% % O = 64 / 120.4 x 100 = 53.2% The percentage composition is 20.2% Magnesium, 26.7% Sulfur and 53.2% Oxygen |
Calculate the percentage composition of CH3Cl
|
M(C) = 12 g/mol
M(H) = 1.01 x 3 = 3.03 g/mol M(Cl) = 35.5 g/mol M(CH3Cl) = 12 + 3.03 + 35.5 = 50.53 g/mol M(CH3Cl) = 50.53 g/mol |
% C = 12 / 50.53 x 100
= 23.7% % H = 3.03 / 50.53 x 100 = 6.00% % Cl = 35.5 / 50.53 x 100 = 70.3% The percentage composition is 23.7% Carbon, 6.00% Hydrogen and 70.3% Chlorine |
What is the percentage composition of sulfuric acid (H2SO4)?
|
M(H) = 1.01 x 2
= 2.02 g/mol M(S) = 32.1 g/mol M(O) = 16 x 4 = 64 g/mol M(H2SO4) = 2.02 + 32.1 + 64 = 98.12 g/mol |
% H = 2.02 / 98.12 x 100
= 2.06% % S = 32.1 / 98.12 x 100 = 32.7% % O = 64 / 98.12 x 100 = 65.2% The percentage composition is 2.06% Hydrogen, 32.7% Sulfur and 65.2% Oxygen |
Empirical formlae
The simplest ratio of atoms in the compound
Calculate the empirical formula of a compound that is 65.5% carbon, 5.5% hydrogen and 29% oxygen by mass
C |
H |
O |
|
m (g) |
65.5 |
5.5 |
29.0 |
n = m / M (mol) |
65.5g / 12g/mol = 5.46 |
5.5g / 1.0 g/mol = 5.5 |
29g / 16g/mol = 1.81 |
Divide by the smallest |
5.46 / 1.18 = 3 |
5.5 / 1.18 = 3 |
1.18 / 1.18 = 1 |
Simplest whole number ratio |
3 |
3 |
1 |
The Empirical Formula is C3H3O
- Convert percentages to masses eg. 65.5% Carbon equates to 65.5 grams
- Calculate the amount of each atom n = m/M eg. n(C) = 65.5g / 12 g/mol
- Divide all amounts by the smallest eg. divide each by 1.18
- Multiply results by a constant to get a whole number ratio - here we already have a whole number ratio
Oxalic acid is 26.7% carbon, 2.2% hydrogen and 71.1% oxygen. Calculate the empirical formula of oxalic acid.
Carbon |
Hydrogen |
Oxygen |
|
m (g) |
26.7 |
2.2 |
71.1 |
n = m / M (mol) |
n = 26.7g / 12g/mol = 2.23 |
n = 2.2g / 1g/mol = 2.2 |
n = 71.1g / 16g/mol = 4.44 |
Divide by the smallest |
2.23 / 2.2 = 1 |
2.2 / 2.2 = 1 |
4.44 / 2.2 = 2 |
Simplest whole number ratio |
1 |
1 |
2 |
Empirical Formula is CHO2
Calculate the empirical formula of a compound containing 51.9% carbon, 4.8% hydrogen, 23.1% oxygen and 20.2% nitrogen
C |
H |
O |
N |
|
m (g) |
51.9 |
4.8 |
23.1 |
20.2 |
n = m/M (mol) |
n = 51.9g / 12g/mol = 4.32 |
n = 4.8g / 1g/mol = 4.8 |
n = 23.1g / 16g/mol = 1.44 |
n = 20.2g / 14g/mol = 1.44 |
Divide by the smallest |
4.32 / 1.44 = 3 |
4.8 / 1.44 = 3.33 |
1.44 / 1.44 = 1 |
1.44 / 1.44 = 1 |
Simplest whole number ratio |
3 x 3 = 9 |
3.33 x 3 = 10 |
1 x 3 = 3 |
1 x 3 = 3 |
Empirical formula = C9H10O3N3
Molecular formula
The Empirical Formula is C3H3O
|
This substance has a molar mass of 55 g/mol. Calculate the molecular formula
You must know or work out the empirical formula before you can do this x = M(molecular formula) / M(empirical formula) = 55g/mol / 55g/mol = 1 Multiply the subscripts in the empirical formula by 1 Molecular formula: C3H3O |
|
Oxalic acid has a molar mass of 90 g/mol, calculate the molecular formula
You must know the empirical formula first x = M(molecular formula) / M(empirical formula) = 90g/mol / 45g/mol = 2 Multiply the subscripts in the empirical formula by 2 Molecular formula: C2H2O4 |
Empirical Formula is CHO2
Vitamin C contains 40.92% carbon, 4.58% hydrogen and 54.5% oxygen. The molar mass of vitamin C is 176 g/mol. Calculate the empirical and molecular formula of vitamin C.
|
The empirical formula for Vitamin C is C3H4O3
x = M(molecular formula) / M(empirical formula) x = 176g/mol / 88g/mol = 2 The molecular formula of Vitamin C is C6H8O6 |